|
Journal of the Chemical Society (Resumed): 3681. 
"The Viscosities of Iodine Pentafluoride and Ditellurium Decafluoride". Journal of the American Chemical Society. "Dielectric Constants of Liquid Chlorine Trifluoride and Iodine Pentafluoride". "Iodine Pentafluoride, Freezing and Boiling Point, Heat of Vaporization and Vapor Pressure-Temperature Relations". 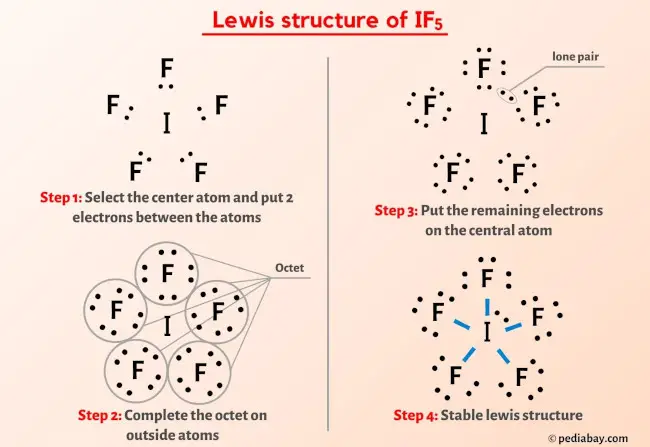
"The Vibrational Spectra and Structures of Iodine Pentafluoride and Heptafluoride". "Rearrangement of Amides with Iodine Pentafluoride". Journal of the Chemical Society: 2789–94. "Preparation and Crystal Structure of Osmium Pentafluoride". Zeitschrift für anorganische und allgemeine Chemie (in German). Zeitschrift für Anorganische und Allgemeine Chemie (in German). "Fluorierung von Verbindungen des Kohlenstoffs (Benzol und Tetrachlormethan mit Jod-5-fluorid, sowie Tetrachlormethan mit Fluor) ". "Crystal structure of Iodine Pentafluoride at -80°". Primary amines react with iodine pentafluoride forming nitriles after hydrolysis References For example, the reduction of osmium hexafluoride to osmium pentafluoride with iodine is conducted in a solution in iodine pentafluoride: 10 OsF 6 + I 2 → 10 OsF 5 + 2 IF 5 It has been used as a solvent for handling metal fluorides. Upon treatment with fluorine, it converts to iodine heptafluoride: IF 5 + F 2 → IF 7 IF 5 reacts vigorously with water forming hydrofluoric acid and iodic acid: This exothermic reaction is still used to produce iodine pentafluoride, although the reaction conditions have been improved. It was first synthesized by Henri Moissan in 1891 by burning solid iodine in fluorine gas. It is used as a fluorination reagent and even a solvent in specialized syntheses. Polarity Polarity is the separation of electric charge on a molecule. It is a colorless liquid, although impure samples appear yellow. Selinium is nonpolar IF5 is polar because all the I-F bonds are polar and. A molecule has a see-saw structure when the central atom is bonded to 4 other atoms and has 1 lone pair.Iodine pentafluoride is an interhalogen compound with chemical formula IF 5. lincoln steffens apush definitionĬorrect Option −D (See-Saw) Reason:- SCl 4 has a lone pair. Iodine Pentafluoride, IF5 Molecular Geometry & Polarity H2O Lewis structure. Linear - $\ce$ orbital produce two hybrid orbitals oriented $180^\circ$ apart. If you know one, then you always know the other. Hybridization was invented to make quantum mechanical bonding theories work better with known empirical geometries.Preparation and structure It is obtained by treating sulfur dichloride with chlorine at 193 K: (1) It melts with simultaneous decomposition above −20 ☌. The corresponding SF 4 is a stable, useful reagent. It has only been obtained as an unstable pale yellow solid. sp, sp2, sp3, sp3d, sp3d2 This problem has been solved! Sulfur tetrachloride is an inorganic compound with chemical formula SCl 4. SCl2, SCl4, SCl6Identify the type of hybridization for thecentral sulfur atom in each compound. Question: What is the hybridization of the central atom in the sulfur tetrachloride (SCIA) molecule? Х $ ?Science Chemistry Chemistry questions and answers Sulfur forms the following compounds withchlorine. (Valence electrons are the number of electrons present in the outermost shell of an atom).You'll get a detailed solution from a subject matter expert that helps you learn core concepts. 
Consequently, N 2 O should have a higher boiling point. Argon and N 2 O have very similar molar masses (40 and 44 g/mol, respectively), but N 2 O is polar while Ar is not. In order to draw the lewis structure of SCl4, first of all you have to find the total number of valence electrons present in the SCl4 molecule. Helium is nonpolar and by far the lightest, so it should have the lowest boiling point. C = Cation charge (positive charge) A= Anion charge (negative charge) Now, on the above hybridization formula, we have to put corresponding values to achieve …Step #1: Calculate the total number of valence electrons Here, the given molecule is SCl4. M = Total number of monoatomic atoms bonded to the central atom. of valence electrons presence on the molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
